

LAS VEGAS, Dec. 6, 2017 /PRNewswire/ -- Today, the PUREVUE™ Visualization System was unveiled at the Orthopaedic Summit 2017: Evolving Techniques. This new offering from DePuy Synthes*, part of the Johnson & Johnson Medical Devices Companies**, is a state-of-the-art imaging platform for minimally invasive endoscopic surgery.
Approximately 18.6 million endoscopic procedures are performed each year in the United States.1 In these procedures, case efficiency and patient outcomes are often linked to how well the surgeon can see the targeted anatomy.
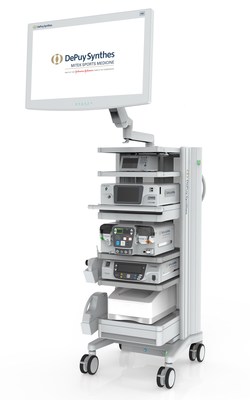
The PUREVUE Visualization System combines a light source, image processer and local image management for a reduced operating room (OR) footprint, and includes intuitive touch-screen navigation and customizable settings for many minimally invasive surgery (MIS) applications. The autoclavable camera head features a proprietary sensor designed for endoscopy, that delivers native HD image acquisition for a clear view throughout the procedure that is further enhanced by multiple monitor options, including ultra-high-definition 4K. The forward-compatible system is also augmented by a wide range of visualization accessories.
"With Americans living longer and seeking shorter recovery times, the call for minimally invasive procedures is accelerating," said Stephanie Chamberlain, Vice President and Global Platform Leader, DePuy Synthes Mitek Sports Medicine. "We are committed to bringing forward innovation that responds to evolving patient needs and the PUREVUE Visualization System is an example of how our advanced technologies enable high quality patient care."
The PUREVUE Visualization System also completes the company's capital equipment offering in arthroscopy, a type of endoscopic surgery focused on joint anatomy, which includes the VAPR VUE™ Radiofrequency System and the FMS VUE™ Fluid Management and Tissue Debridement System. Together, these solutions bolster Mitek Sport Medicine's portfolio of implants and instrumentation used for a wide spectrum of arthroscopic soft tissue procedures.
About DePuy Synthes
DePuy Synthes, part of the Johnson & Johnson Medical Devices Companies, provides one of the most comprehensive orthopaedics portfolios in the world. DePuy Synthes solutions, in specialties including joint reconstruction, trauma, craniomaxillofacial, spinal surgery and sports medicine, are designed to advance patient care while delivering clinical and economic value to health care systems worldwide. For more information, visit www.depuysynthes.com.
1 Perry, Anne F., et al. "Burden of Gastrointestinal Disease in the United States: 2012 Update" Gastroenterology. 2012 November; 143(5): 1179–1187.e3. doi:10.1053/j.gastro.2012.08.002 Update.
*DePuy Synthes represents the products and services of DePuy Synthes, Inc. and its affiliates.
**The Johnson & Johnson Medical Devices Companies comprise the surgery, orthopaedics, and cardiovascular businesses within Johnson & Johnson's Medical Devices segment.
©DePuy Synthes 2017. All rights reserved.
DSUS/MTK/1117/1100(1)
December 2017

SOURCE DePuy Synthes